You are here: / Home
Influenced melting point of water by magnetic field
Controlled sub-cooling of products in freezing processes
The aim of the research project is to find a solution that enables the freezing processes of aqueous systems to be influenced and controlled in a targeted manner using magnetic fields. Publications suggest that the melting point of water in the magnetic field is affected [1]. As part of the research project, an experimental apparatus is planned and set up to allow systematic investigations of the melting point of water and aqueous solutions at different magnetic field strengths.
The intention is to cool water-containing systems below the "normal" freezing point at 0 °C within a magnetic field, regardless of the nucleation that occurs, and then to freeze them by switching off the magnetic field.
The cause of the expected change in the melting point / freezing point by the magnetic field is assumed to be the weakening of the hydrogen bonds in the water in the magnetic field.
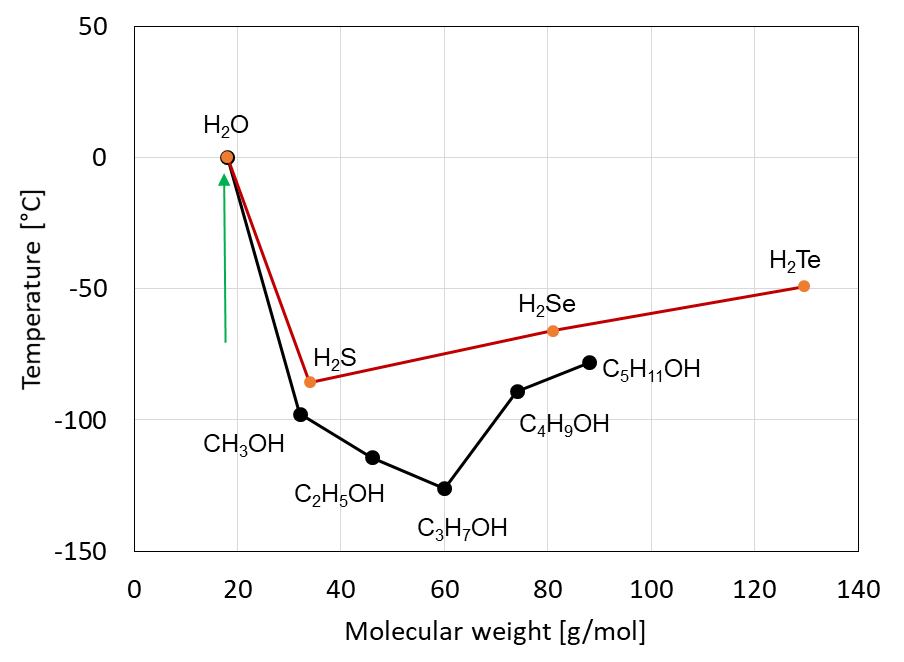
There is a wide range of anomalous properties of water caused by hydrogen bonds. In Figure 1 this is shown for the example of the melting points / freezing points for hydrogen compounds of the elements in the 6th main group of the periodic table of the chemical elements as well as for alcohols (including water) within their homologous series. A significant deviation from the general trend can be seen for water with its strong hydrogen bonds.
A potential practical application of the magnetic field controlled reduction of the freezing point of water is the improvement of the product quality of frozen food.
References:
[1] L. Otero, A. C. Rodriguez, M. Pérez-Mateos und P. D. Sanz, „Effects of magnetic fields on freezing: application to biological products“, Food Science and Food Safety, Vol. 15; 2016.